News Story
UMD Researchers Discover Methods to Mediate Membrane-Nanoparticle Interactions

A team of graduate students, led by the Mechanical Engineering Assistant Professor Siddhartha Das, has published findings on how electrostatics control interactions between the membrane and nanoparticle in Applied Physics Letters. Ph.D. students Shayandev Sinha and Haoyuan Jing and M.S. student Harnoor Singh Sachar were co-authors on this publication.
This research has the potential to improve the targeted drug delivery process in the medical field. An example of this process would be in cancer treatments, where Das and his student’s theory of controlling interactions would allow nanoparticle-based drug delivery to occur on the affected cells more effectively.
Understanding the interactions between nanoparticles (NPs) and the cellular plasma membrane (PM) is vital for using NPs for targeted drug and gene delivery, imaging of cellular processes, and many other applications. Such PM-NP interactions typically occur through the chemical interactions between the ligands attached on the NPs and the receptors emerging from the PM surface. Such receptor-ligand (RL) interactions ensure the necessary specificity in the membrane-NP interactions that is vital for precisely targeted drug delivery.
To trigger this RL interaction, one must ensure that the NP approaches the PM to a minimum distance of separation (DOS) of dRL, which denotes the length of the R-L complex.
In this study, Das and his team shed light on the detailed electrostatics-mediated mechanism that dictate the critical condition that will ensure the onset of the R-L-mediated PM-NP interactions. The team considered a gold NP approaching a negatively charged PM. The PM induced a negative zeta potential at the surface of the NP, which in turn sets up a strong electrostatic repulsion between the NP and the membrane. This repulsion, which drives the NP away from the membrane, is countered by the attractive influences of the thermal fluctuations and the van der Waals (vdW) interactions.
Under such conditions, Das and his team determined that the repulsive and attractive influences balance each other at membrane-NP DOS of dg,c,1. For any DOS less than dg,c,1, the repulsive effects overcome the attractive influences enforcing the NP to be driven away from the membrane. This scenario, as proposed by Das, helps to pinpoint a design criterion for the length of the ligand (or equivalently, the length dRL of the R-L complex) – this study proposes that in order to ensure the R-L mediated interactions, dRL should be such that it is always much larger than dg,c,1. This study provides a previously unknown standard for designing ligands on NPs that help them to overcome the electrostatic barrier for interacting with the PM through these R-L interactions.
Of the findings, Das notes “This design information will be quintessential for the designing of novel NP-based drug delivery mechanisms by exploiting electrostatic effects in manner that has remained very poorly explained.”
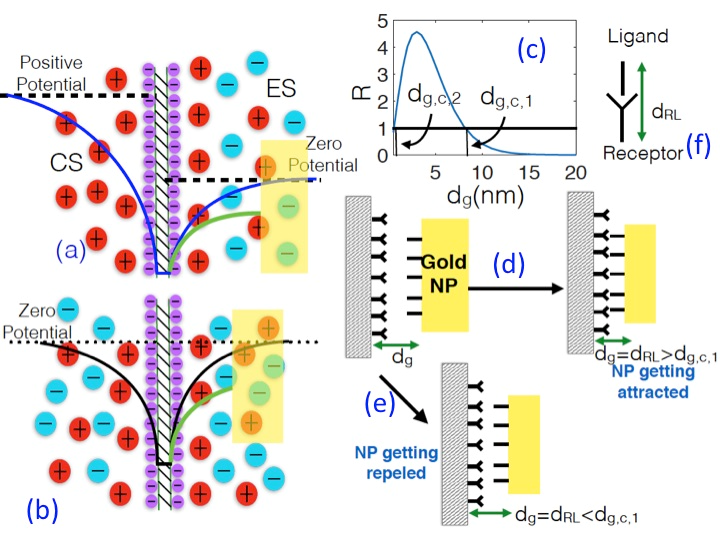
Schematic of the EDL electrostatics for (a) a semi-permeable membrane and (b) a fully permeable membrane. For both the cases, an approaching gold NP (from the electrolyte side) attains a negative potential on its surface, which repels it from the membrane. (c) Variation of the dimensionless energy ratio R [where R=Eelec/(EvdW+kBT)] as a function of the separation distance dg between the membrane and the NP. The attractive influences (EvdW+kBT) can overcome repulsion energy (Eelec) only when dg>dg,c,1. Hence, a R-L interaction becomes possible (such an interaction is necessary for the NP-membrane binding) for dRL>dg,c,1 (d), while the NP is driven away from the membrane for dRL<dg,c,1 (e). (f) Schematic of the RL bond and quantification of the R-L length dRL. This figure and the caption has been taken from Ref. 1.
Reference
Reprinted with permission from "S. Sinha. H. Jing, H. S. Sachar, and S. Das, “Role of plasma membrane surface charges in dictating the feasibility of membrane nanoparticle interactions”, Appl. Phys. Lett., Vol. 111, 263702 (2017)." Copyright 2017, AIP Publishing LLC. This article may be downloaded for personal use only. Any other use requires prior permission of the author and the AIP Publishing LLC."
Published January 16, 2018













