News Story
ME Researchers Develop Methods to Mediate Nanoparticle Adhesion

A team of graduate students, led by the Mechanical Engineering Assistant Professor Siddhartha Das, has published findings on how electrostatics can ensure non-specific nanoparticle (NP) adhesion to stiffer cell membranes in Applied Physics Letters [1]. Ph.D. students Shayandev Sinha, Haoyuan Jing and M.S. student Harnoor Singh Sachar were co-authors on this publication.
This research has the potential to improve the drug delivery process to cells that have become stiffened due to certain pathological and/or physiological conditions and are not treatable by standard NP-based drug delivery approaches.
Some examples of such stiffened cells are: blood cells (RBCs) in presence of oxidative stresses, RBCs demonstrating disorders in cytoskeletal activities, cell stiffened due to the actin recruitment in the presence of external stresses, and many more. Also, such adhesion property can develop biomimetic nanoparticles where the nanoparticles may be coated with a layer of lipid bilayer stiffened by the presence of excessive cholesterol.
This research stems out of non-specific adhesion, where the nanoparticles interact with cell membranes through physical contact without involving receptor-ligand interactions.
Das and his students establish that for a metal NP undergoing such physical, non-specific adhesion to the cell membrane, there is a joint destruction of the effect of the membrane surface charge as well as the charge cloud (known as the electric double layer or EDL) that the membrane charge induces.
The destruction of the membrane surface charge makes the overall energy change negative (or favorable), while the destruction of the EDL makes the energy change positive (or unfavorable). These two effects compete with each other and for a metal NP, the extent of the destruction of the effect of the membrane surface charge is large enough to ensure that the net energy change for the adhesion process is negative. This energy gained ensures NP non-specific adhesion to stiffer membranes (or membrane with larger bending modulus B). Figure 1 summarizes this discovered phenomenon schematically.
Of the findings, Das says “This design information will allow us more flexibility in treating cells with larger stiffness and would also open up the door to better manipulate cell electrostatics for other applications.”
Reference
[1] S. Sinha. H. Jing, H. S. Sachar, and S. Das, “Surface charges promote nonspecific nanoparticle adhesion to stiffer membranes”, Appl. Phys. Lett., Vol. 112, 163702 (2018).
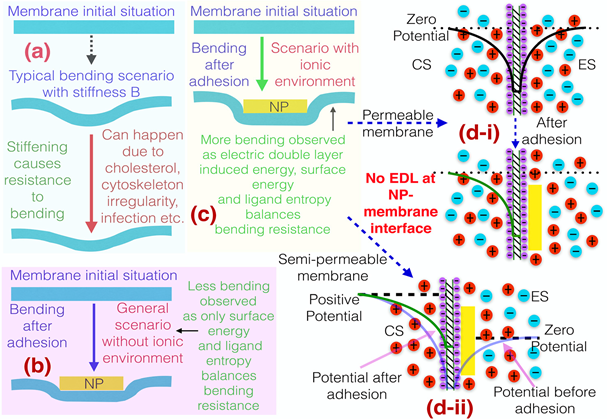
Figure 1: Schematic depicting the surface charge mediated NSA of NPs to stiffer membranes. (a) Description of the bending and the role of the bending modulus B. (b) NP NSA without electrostatic effects—absence of any ionic condition will lead to the NP NSA and hence bending of a less PM. (c) NP NSA (non-specific adhesion) in the presence of the electrostatic effects (i.e., membrane surface charges and the resulting EDL) – presence of the ionic condition will lead to the NP NSA and hence bending of a much stiffer PM. (d-i) NSA of the NP to the fully permeable plasma membrane – ion and w distribution before [see top of (d-i)] and after [see bottom of (d-i)] the NP adhesion. (d-ii) NSA of the NP to the semi-permeable plasma membrane and the ion and w distribution before and after the adhesion. The figure and the caption has been reproduced from Ref. 1 with permission.
Published May 11, 2018















